Non-destructive battery test using special nuclear magnetic resonance method
07/05/2026
Rechargeable batteries are everywhere, from portable electronic devices and electric vehicles to renewable energy storage. Battery failures are often due to the loss or chemical decomposition of the electrolyte. An international research team with the participation of the Helmholtz Institute Mainz (HIM), a branch office of the GSI Helmholtz Centre for Heavy Ion Research in Darmstadt, Germany, the Johannes Gutenberg University Mainz (JGU), the Physikalisch-Technische Bundesanstalt in Berlin, Germany, and New York University, USA, has now dealt with the question of how to enable a non-destructive diagnosis of the electrolyte through the battery housing using special nuclear magnetic resonance procedures[1].How does a rechargeable battery work? A battery stores electrical energy in chemical form. Inside are two metallic electrodes and a medium called an electrolyte. During discharge, chemical reactions occur in which charged particles migrate inside, while electrons flow over the outer circuit, thus providing electrical energy. This process can be reversed in the battery: the charging resets the chemical processes, so that the energy storage can be used again. Over many charging cycles, the electrolyte changes, ages or may leak, which can lead to unusability or, in the worst case, even a hazard from heat development or explosion.
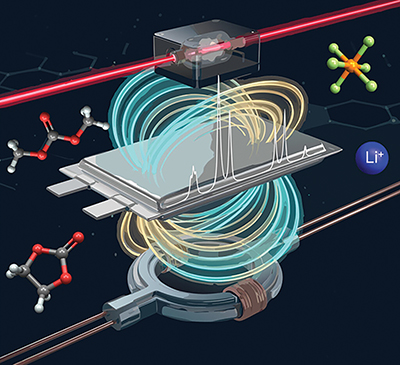 | ||
| Graphical representation of the ZULF-NMR measurement of a pouch cell (centre) using quantum sensors such as optically pumped magnetometers ((OPMs), above) and superconducting quantum interference devices ((SQUIDs), below) that can detect and quantify the low magnetic fields generated by the nuclear spins of the molecules in the battery electrolyte Photo courtesy of F Teleanu and A Fabricant, using the Global Partnership on Artificial Intelligence (GPAI) |
“Reliable methods of testing the condition of the battery non-destructively are missing, since the amount and chemical composition of the electrolyte cannot be determined through the housing using conventional techniques. This is exactly where our research comes in,” said Dr Anne Fabricant, who was involved in the experiments at HIM and the Physikalisch-Technische Bundesanstalt in Berlin. “We study the batteries using the so-called zero-field and ultra-low-field magnetic resonance. For this technology, the housings are transparent, so we can look inside.” In this diagnostic method, also called zero-to-ultra-low-field nuclear magnetic resonance (ZULF-NMR), nuclear spin resonance is measured without the action of a strong external magnetic field.
“In our tests, we were able to show the direct detection and quantification of both the solvent and lithium salt components of commercial electrolytes through metal battery housings,” explained Professor Dmitry Budker, who works at HIM and JGU and is one of the pioneers of the ZULF-NMR process. “These were realistically packaged battery cells, also so-called pouch cell geometries, such as those used for electric vehicles. In doing so, we have demonstrated the feasibility and paved the way for a practical application of the technology.”
In the future, ZULF-NMR could be used to test rechargeable batteries for their integrity during operation in the context of operando measurements. This is an increasingly important topic because they are used in many places, for example in small mobile devices such as mobile phones or notebooks, but also on a large scale in electric vehicles. They are particularly relevant in the storage of renewable energies. In addition, the measurements provide a deeper understanding of electrochemical processes and the development of next-generation battery cell technologies.
“The ability to characterise electrolyte volumes and compositions non-destructively supports optimal battery design and serves as an important tool for quality control throughout the entire lifecycle of a cell,” said Professor Alexej Jerschow of New York University, cooperation partner and recipient of the Carl Zeiss Humboldt Research Award.
The research team led by Professor Budker is planning further experiments to improve diagnostics. “We still have a lot of ideas on how to make the detection more accurate and faster, how we can study larger batteries or how the method can be made more cost-efficient,” said Professor Budker. “I am convinced that the technique will find a place in the long term alongside other, more invasive diagnostic methods.”
www.uni-mainz.de
budker.uni-mainz.de
Reference
1. A M Fabricant et al, ‘Enabling non-destructive observation of electrolyte composition in batteries with ultra-low-field nuclear magnetic resonance’, Chemical Science, Vol 17, No 12, pp 5877-5887, 2026. DOI: 10.1039/D5SC04419G